

Revolutionized is reader-supported. When you buy through links on our site, we may earn an affiliate commision. Learn more here.
Histological analysis is a cornerstone of modern scientific investigation, enabling researchers to examine tissue architecture and cellular detail at microscopic resolution. It uncovers structural changes that signal disease progression and developmental patterns. This microscopic tissue evaluation supports breakthroughs in oncology and regenerative medicine.
In biotechnology and pharmaceutical research, it strengthens drug development pipelines by revealing toxicity and cellular repair mechanisms. Meanwhile, advancements in high-resolution imaging and digital pathology platforms reshape traditional laboratory workflows. Computational pattern recognition and data integration tools now enhance reproducibility and elevate histological analysis into a faster, more scalable discipline.
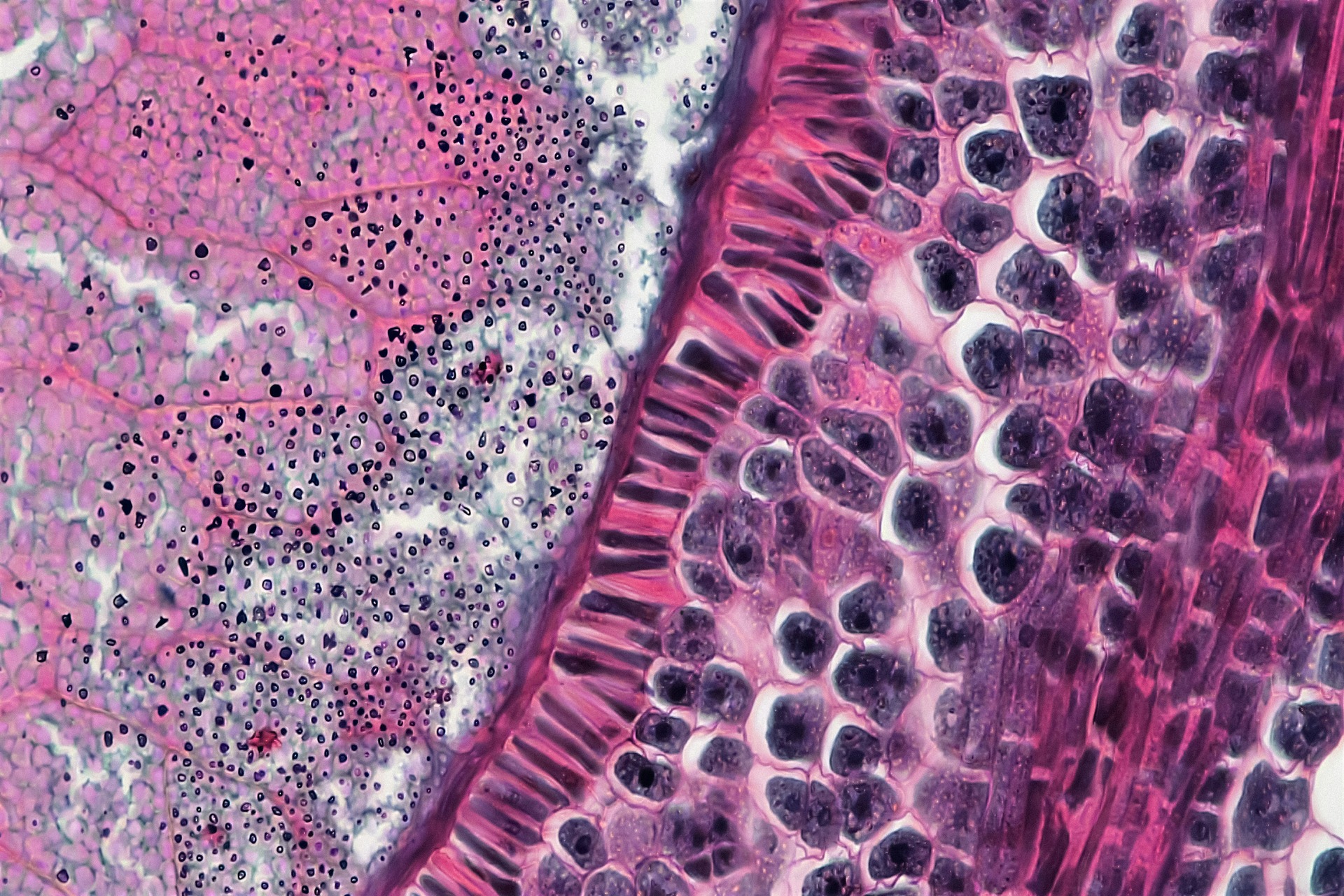
Histological analysis is the microscopic examination of biological tissues designed to reveal their structural composition and functional organization. It uncovers how cells are arranged within tissues and how those arrangements shift in response to injury or therapeutic intervention. Through careful fixation, sectioning and staining, it produces detailed visual evidence that allows pathologists to study the alterations in tissue architecture caused by various diseases.
Changes in cell morphology and disrupted structural integrity often provide early indicators of degeneration or inflammation. This method holds critical importance across pathology and neuroscience, where it informs tumor grading and validates engineered tissues. By connecting microscopic structure to clinical outcomes, histological analysis shapes scientific discovery and modern medical practice.
Understanding histological analysis requires a closer look at the core techniques that make microscopic evaluation possible. Each method plays a distinct role in preserving tissue integrity and revealing the structural details that drive accurate interpretation.
Microtomes create ultra-thin tissue slices by advancing a fixed specimen toward a finely sharpened blade, producing sections often just a few micrometers thick. Mastery of precise sectioning also reveals the complexities of cellular composition and function, which drives meaningful advances in medical research. Even slight inconsistencies in thickness can distort morphology, making precision essential for accurate microscopic evaluation and reliable interpretation.
Paraffin sectioning involves embedding tissue in wax after fixation and dehydration, resulting in highly stable samples suited for detailed structural analysis. Frozen sectioning, by contrast, preserves tissue through rapid freezing and allows faster preparation. Each approach offers distinct advantages depending on the research goal and level of structural detail required.
Staining binds specific dyes to cellular components, making otherwise transparent structures visible under a microscope. Without staining, many tissue elements appear nearly colorless, which limits the ability to distinguish nuclei and extracellular structures. By selectively highlighting these features, staining allows clearer interpretation of morphology and pathological change.
Beyond routine assessment, special stains target specific biological components. Techniques such as Masson’s trichrome highlight collagen fibers, and Gram staining identifies microorganisms within infected tissues. These targeted methods reveal details that standard staining cannot capture.
Immunohistochemistry uses antibodies to detect specific proteins within tissue sections, allowing highly targeted molecular visualization. Antibodies are crucial proteins produced by the immune system to attack and fight off foreign toxins that enter the body. When tagged with visible labels, these antibodies reveal the location and abundance of proteins directly within preserved tissue architecture.
Compared with traditional staining methods that primarily highlight structural features, immunohistochemistry delivers molecular-level insight. It reduces diagnostic ambiguity and strengthens the link between microscopic observation and personalized treatment strategies.
Evaluating the effectiveness of histological analysis requires examining its diagnostic power and research impact. Its value becomes clear when microscopic insight translates into accurate clinical decisions and measurable scientific progress.
Histological analysis reveals how experimental compounds affect real tissue structures at the cellular level. Researchers rely on it to detect inflammation and subtle morphological changes that signal adverse reactions before clinical symptoms appear. Histological methods are also essential for cell removal efficiency evaluation and ultrastructure preservation analysis, particularly in biomaterials research and tissue engineering, where structural integrity determines functional success.
By linking microscopic structure to therapeutic response, histological analysis supports the advancement of personalized medicine. Protein expression patterns and tissue-specific biomarkers guide more precise treatment strategies tailored to individual patients.
Histological analysis detects infections and inflammatory diseases by revealing structural abnormalities at the cellular level. Malignant tumors often display disrupted architecture and invasive growth patterns, while infections and inflammatory conditions produce distinct cellular infiltrates. These visible changes provide immediate diagnostic clues that guide further testing and treatment planning.
When compared with molecular diagnostics, histology offers contextual insight that genetic or proteomic data alone cannot provide. However, accurate interpretation depends heavily on trained expertise. Skilled pathologists integrate staining characteristics and clinical history to deliver reliable diagnoses, reinforcing the human judgment that remains essential in histological analysis.
Histological analysis delivers exceptionally high-resolution structural detail, allowing clinicians and researchers to observe subtle architectural changes that signal disease progression. However, the preparation process remains time-intensive, requiring careful sectioning and staining before evaluation can begin. In fact, a 2020 study in England revealed that 44% of the National Health Service’s clinical trials contained impossible figures and incorrect calculations.
This number underscores the broader risks of analytical error in complex research systems. Variability in staining intensity and human interpretation can further influence outcomes. Differences in technique or subjective judgment may lead to interobserver variability, reinforcing the need for standardized protocols and complementary digital validation tools.
The rise of automated slide scanning has transformed traditional microscopy into a digital workflow capable of storing and analyzing entire tissue sections at high resolution. Whole-slide imaging platforms now convert glass slides into data-rich files that can be shared and reviewed remotely.
Machine learning algorithms build on this foundation by quantifying staining intensity and flagging abnormalities across thousands of images. These systems help address cognitive bias, which can be a source of error in diagnostic pathology, especially in tumor grading, where subtle differences influence clinical decisions. By providing objective measurements and decision support, AI tools reduce variability between observers.
Benefits include streamlined case prioritization and reduced human error in repetitive evaluation tasks. However, ethical considerations remain critical, including transparency in algorithm design and rigorous validation requirements before clinical deployment. Responsible integration ensures that digital pathology strengthens expert judgment rather than replacing it.
Emerging multiplex imaging platforms now enable the simultaneous detection of multiple biomarkers within a single tissue section. By labeling different proteins with distinct fluorescent tags, researchers can map complex cellular interactions and better understand tumor microenvironments. This layered insight moves beyond single-marker analysis and captures the dynamic biology occurring within intact tissue.
Techniques such as tissue clearing and advanced 3D microscopy allow volumetric tissue analysis, revealing structural relationships across entire organs rather than thin slices alone. Three-dimensional reconstruction provides context for neural mapping and engineered tissue integration.
Histological analysis combines foundational techniques with advanced imaging to deliver reliable diagnostic and research insight. Ongoing innovation in automated slide scanning and AI-driven evaluation improves consistency and analytical depth. Despite rapid advances in molecular technologies, histological analysis remains central to scientific discovery by linking cellular structure to real-world biological outcomes.
Revolutionized is reader-supported. When you buy through links on our site, we may earn an affiliate commision. Learn more here.


This site uses Akismet to reduce spam. Learn how your comment data is processed.